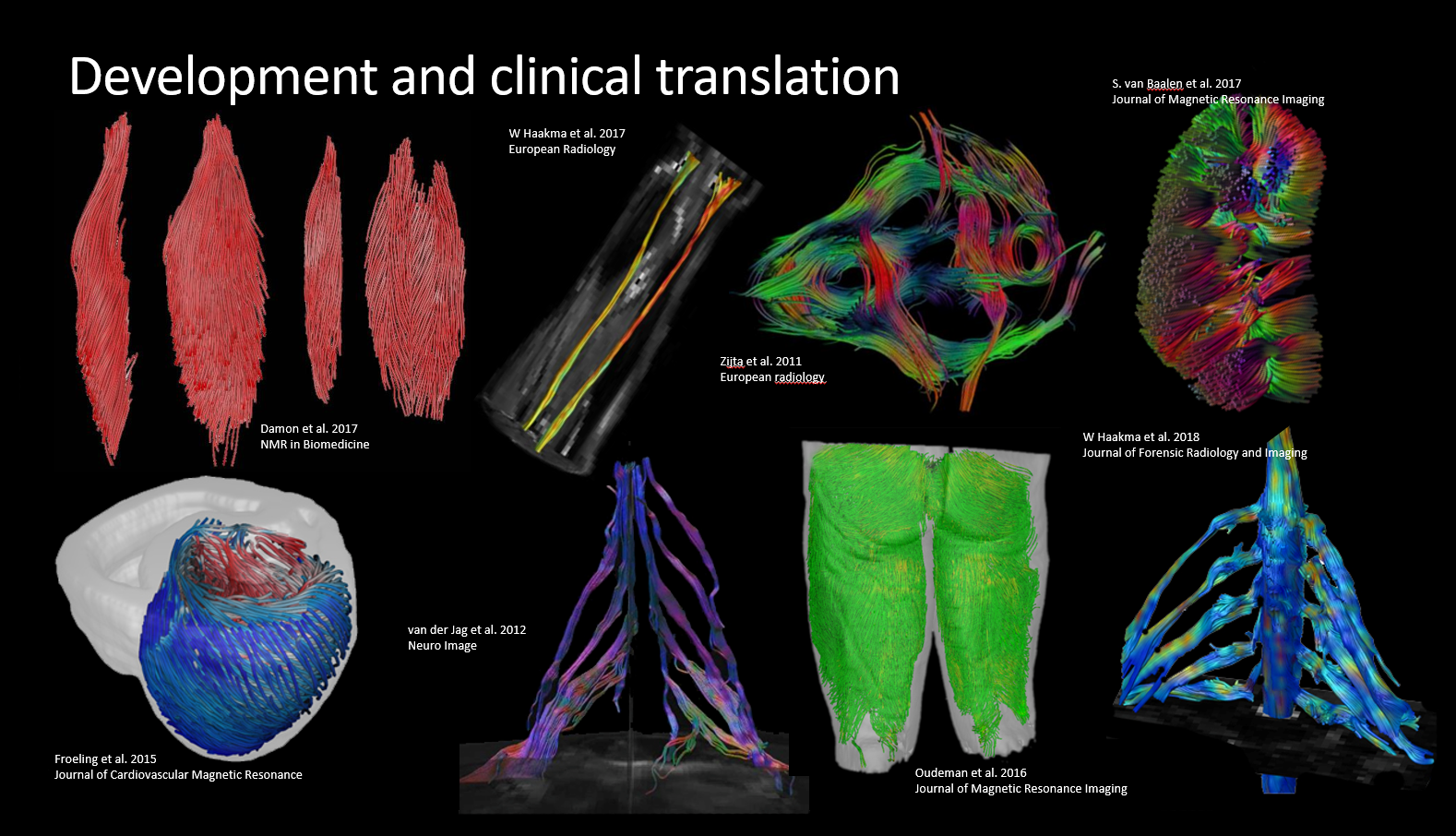
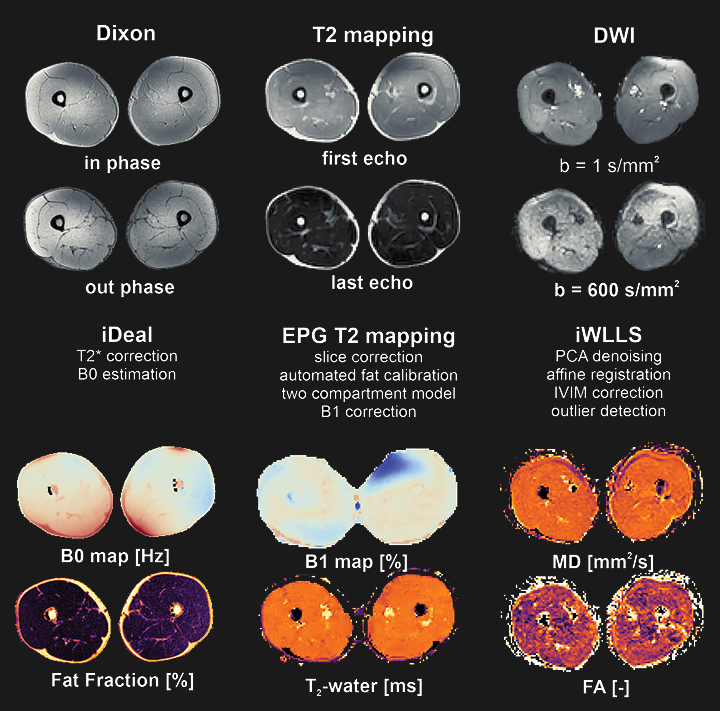
a little bit of history
About QMRITools
About QMRITools
QMRITools is a comprehensive collection of tools and functions designed for processing quantitative MRI data. Developed specifically for the Wolfram language, QMRITools operates within the latest version of Wolfram Mathematica. While it does not offer a graphical user interface (GUI), its primary objective is to enable rapid batch data processing and to facilitate the development and prototyping of new functions. At its core, the toolbox includes a variety of functions for data manipulation and restructuring.
Key Features of QMRITools
Froeling et al. 2012- Extensive Function Library: QMRITools boasts over 450 custom functions, encompassing more than 30,000 lines of code, complete with over 750 documentation pages and demonstrations.
- Wide Range of Applications: Initially developed for analyzing DWI data of muscle, QMRITools has expanded to support cardiac analysis (including tagging and T1 mapping), Dixon reconstruction, EPG modeling and fitting, j-coupling simulations, and more.
Applications in Quantitative MRI
QMRITools was primarily developed for use in quantitative muscle MRI (Froeling et al. 2012), nerve imaging, and cardiac magnetic resonance imaging. The toolbox evolves continuously, with new functions added in response to ongoing research needs.
Recognition and Awards
QMRITools received the “Best Open Source Tool Award” from the Quantitative MRI Study Group during the 2023 ISMRM conference in Toronto. This accolade highlights the toolbox’s significant contributions to the field of quantitative MRI. Additionally, it was awarded the Wolfram Innovator Award in 2023.
Learning and Engagement
To gain deeper insights into QMRITools, you can watch a live discussion featuring the Wolfram Academic Outreach Team, which delves into the role of computational Wolfram technology in the toolbox’s development. Additionally, a detailed presentation on the paclet functionality was shared with the Wolfram R&D Team, providing an in-depth look at the toolbox’s capabilities.
Referencing
When using the toolbox it is appreciated if you cite one of the following articles:
- Froeling M: QMRTools: a Mathematica toolbox for quantitative MRI analysis. J Open Source Softw 2019; 4:1204. link
- Froeling M, et al.: Reproducibility of diffusion tensor imaging in human forearm muscles at 3.0 T in a clinical setting. Magn Reson Med 2010; 64:1182-1190. link
- Froeling M, et al.: Diffusion-tensor MRI reveals the complex muscle architecture of the human forearm. J Magn Reson Imaging 2012; 36:237-248. link
- Schlaffke L, et al.: Multi‐center evaluation of stability and reproducibility of quantitative MRI measures in healthy calf muscles. NMR Biomed. 2019;32:e4119 link
Publications using QMRITools
1. Mokkenstorm MJK, Monte JR, Suskens JJM, et al.: Diffusion tensor imaging has prognostic value on return to play in hamstring injuries: A prospective cohort study. European Journal of Radiology 2025; 191:112350.
2. Istomina AV, Faber A, Manzhurtsev AV, Ublinskiy MV, Arsalidou M: Neural correlates of solving arithmetic problems in adults. Neuroscience and Behavioral Physiology 2025; 55:905–922.
3. Heskamp L, Schlaffke L, Forsting J, Sleutjes BTHM, Goedee HS, Froeling M: Fasciculation distribution in a healthy population assessed with diffusion tensor imaging. Physiological Reports 2025; 13:e70247.
4. Mathew E, Dortch R, Damon B, Ragunathan S, Quarles CC: Repeatability of diffusion kurtosis tensor parameters in muscles of the lower legs. Magnetic Resonance in Medicine 2025; 93:1306–1313.
5. Gursan A, Kahraman-Agir B, Gosselink M, et al.: Development of a Double Tuned 2H/31P Whole-Body Birdcage Transmit Coil for 2H and 31P MR Applications From Head to Toe at 7 T. NMR in Biomedicine 2025; 38:e5325.
6. Rauh SS, Cameron D, Gurney-Champion OJ, et al.: Investigating skeletal muscle micro-trauma with time-dependent diffusion and the random permeable barrier model. Scientific Reports 2024; 14:31998.
7. Forsting J, Rehmann R, Rohm M, et al.: Prospective longitudinal cohort study of quantitative muscle magnetic resonance imaging in a healthy control population. NMR in Biomedicine 2024; 37:e5214.
8. Forsting J, Wächter M, Froeling M, et al.: Quantitative muscle magnetic resonance imaging in limb‐girdle muscular dystrophy type R1 (LGMDR1): A prospective longitudinal cohort study. NMR in Biomedicine 2024; 37:e5172.
9. Güttsches A, Forsting J, Kneifel M, et al.: Pre‐ and post‐skeletal muscle biopsy quantitative magnetic resonance imaging reveals correlations with histopathological findings. European Journal of Neurology 2024; n/a(n/a):e16479.
10. Froeling M, Heskamp L: The effect of fat model variation on muscle fat fraction quantification in a cross‐sectional cohort. NMR in Biomedicine 2024; 37:e5217.
11. Schlaffke L, Rehmann R, Froeling M, et al.: Quantitative muscle MRI in sporadic inclusion body myositis (sIBM): A prospective cohort study. Journal of Neuromuscular Diseases 2024; 11:997–1009.
12. Schlaffke L, Rehmann R, Güttsches A-K, et al.: Evaluation of Neuromuscular Diseases and Complaints by Quantitative Muscle MRI. Journal of Clinical Medicine 2024; 13:1958.
13. Winter L, Periquito J, Kolbitsch C, et al.: Open-source magnetic resonance imaging: Improving access, science, and education through global collaboration. NMR in Biomedicine 2024; 37:e5052.
14. Rohm M, Russo G, Helluy X, et al.: Muscle diffusion MRI reveals autophagic buildup in a mouse model for Pompe disease. Scientific Reports 2023; 13:22822.
15. Monte JR, Hooijmans MT, Froeling M, et al.: Diffusion tensor imaging and quantitative T2 mapping to monitor muscle recovery following hamstring injury. NMR in Biomedicine 2023; 36:e4902.
16. Suskens JJM, Secondulfo L, Kiliç Ö, et al.: Effect of two eccentric hamstring exercises on muscle architectural characteristics assessed with diffusion tensor <scp>MRI</scp>. Scandinavian Journal of Medicine & Science in Sports 2022; 33:393–406.
17. Wildenberg L van den, Gursan A, Seelen LWF, et al.: In vivo phosphorus magnetic resonance spectroscopic imaging of the whole human liver at 7 T using a phosphorus whole‐body transmit coil and 16‐channel receive array: Repeatability and effects of principal component analysis‐based denoising. NMR in Biomedicine 2022; 36:e4877.
18. Forsting J, Rohm M, Froeling M, et al.: Quantitative muscle MRI captures early muscle degeneration in calpainopathy. Scientific Reports 2022; 12:1–10.
19. Waterval NFJ, Meekes VL, Hooijmans MT, et al.: The relationship between quantitative magnetic resonance imaging of the ankle plantar flexors, muscle function during walking and maximal strength in people with neuromuscular diseases. Clinical Biomechanics 2022; 94:105609.
20. Forsting J, Rehmann R, Rohm M, et al.: Robustness and stability of volume‐based tractography in a multicenter setting. NMR in Biomedicine 2022; 35:0–3.
21. Hooijmans MT, Habets LE, Berg‐Faay SAM van den, et al.: Multi‐parametric quantitative magnetic resonance imaging of the upper arm muscles of patients with spinal muscular atrophy. NMR in Biomedicine 2022; 35:e4696.
22. Habets LE, Bartels B, Asselman F-L, et al.: Magnetic resonance reveals mitochondrial dysfunction and muscle remodelling in spinal muscular atrophy. Brain 2021; 145:1422–1435.
23. Rehmann R, Schneider-Gold C, Froeling M, et al.: Diffusion Tensor Imaging Shows Differences Between Myotonic Dystrophy Type 1 and Type 2. Journal of Neuromuscular Diseases 2021; 8:949–962.
24. Gursan A, Froeling M, Hendriks AD, et al.: Residual quadrupolar couplings observed in 7 Tesla deuterium MR spectra of skeletal muscle. Magnetic Resonance in Medicine 2021; 87:1165–1173.
25. Rohm M, Markmann M, Forsting J, Rehmann R, Froeling M, Schlaffke L: 3D Automated Segmentation of Lower Leg Muscles Using Machine Learning on a Heterogeneous Dataset. Diagnostics 2021; 11:1747.
26. Forsting J, Rohm M, Froeling M, et al.: High Inter-Rater Reliability of Manual Segmentation and Volume-Based Tractography in Healthy and Dystrophic Human Calf Muscle. Diagnostics 2021; 11:1521.
27. Güttsches A-K, Rehmann R, Schreiner A, et al.: Quantitative Muscle-MRI Correlates with Histopathology in Skeletal Muscle Biopsies. Journal of Neuromuscular Diseases 2021; 8:669–678.
28. Froeling M, Prompers JJ, Klomp DWJ, Velden TA van der: PCA denoising and Wiener deconvolution of 31P 3D CSI data to enhance effective SNR and improve point spread function. Magnetic Resonance in Medicine 2021; 85:2992–3009.
29. Gosselink M, Hoogduin H, Froeling M, Klomp DWJ: No need to detune transmitters in 32‐channel receiver arrays at 7 T. NMR in Biomedicine 2021; 34:e4491.
30. Mazzoli V, Moulin K, Kogan F, Hargreaves BA, Gold GE: Diffusion Tensor Imaging of Skeletal Muscle Contraction Using Oscillating Gradient Spin Echo. Frontiers in Neurology 2021; 12(February):608549.
31. Otto LAM, Froeling M, Eijk RPA van, et al.: Quantification of disease progression in spinal muscular atrophy with muscle MRI—a pilot study. NMR in Biomedicine 2021; 34:e4473.
32. Straatman HCH, Schoor I van der, Froeling M, Van Steenkiste G, Holtackers RJ, Delhaas T: Quantitative Interpretation of Myocardial Fiber Structure in the Left and Right Ventricle of an Equine Heart Using Diffusion Tensor Cardiovascular Magnetic Resonance Imaging. In Lecture Notes in Computer Science (including subseries Lecture Notes in Artificial Intelligence and Lecture Notes in Bioinformatics). Volume 12738 LNCS. Springer; 2021:178–188.
33. Stouge A, Khan KS, Kristensen AG, et al.: MRI of Skeletal Muscles in Participants with Type 2 Diabetes with or without Diabetic Polyneuropathy. Radiology 2020; 297:608–619.
34. Forsting J, Rehmann R, Rohm M, Froeling M, Schlaffke L: Evaluation of interrater reliability of different muscle segmentation techniques in diffusion tensor imaging. NMR in Biomedicine 2020; 34:e4430.
35. Secondulfo L, Ogier AC, Monte JR, et al.: Supervised segmentation framework for evaluation of diffusion tensor imaging indices in skeletal muscle. NMR in Biomedicine 2020; 34:e4406.
36. Hooijmans MT, Froeling M, Koeks Z, et al.: Multi‐parametric MR in Becker muscular dystrophy patients. NMR in Biomedicine 2020; 33:e4385.
37. Otto LAM, Pol W van der, Schlaffke L, et al.: Quantitative MRI of skeletal muscle in a cross‐sectional cohort of patients with spinal muscular atrophy types 2 and 3. NMR in Biomedicine 2020; 33:e4357.
38. Rehmann R, Froeling M, Rohm M, et al.: Diffusion tensor imaging reveals changes in non‐fat infiltrated muscles in late onset Pompe disease. Muscle & Nerve 2020; 62:541–549.
39. Keene KR, Beenakker JM, Hooijmans MT, et al.: T2 relaxation‐time mapping in healthy and diseased skeletal muscle using extended phase graph algorithms. Magnetic Resonance in Medicine 2020; 84:2656–2670.
40. Hooijmans MT, Monte JRC, Froeling M, et al.: Quantitative MRI Reveals Microstructural Changes in the Upper Leg Muscles After Running a Marathon. Journal of Magnetic Resonance Imaging 2020; 52:407–417.
41. Oudeman J, Eftimov F, Strijkers GJ, et al.: Diagnostic accuracy of MRI and ultrasound in chronic immune-mediated neuropathies. Neurology 2020; 94:e62–e74.
42. Forsting J, Rehmann R, Froeling M, Vorgerd M, Tegenthoff M, Schlaffke L: Diffusion tensor imaging of the human thigh: Consideration of DTI-based fiber tracking stop criteria. Magnetic Resonance Materials in Physics, Biology and Medicine 2019; 33:343–355.
43. Monte JR, Hooijmans MT, Froeling M, et al.: The repeatability of bilateral diffusion tensor imaging (DTI) in the upper leg muscles of healthy adults. European Radiology 2019; 30:1709–1718.
44. Schlaffke L, Rehmann R, Rohm M, et al.: Multi‐center evaluation of stability and reproducibility of quantitative MRI measures in healthy calf muscles. NMR in Biomedicine 2019; 32:e4119.
45. Froeling M: QMRTools: A Mathematica toolbox for quantitative MRI analysis. Journal of Open Source Software 2019; 4:1204.
46. Voskuilen L, Mazzoli V, Oudeman J, et al.: Crossing muscle fibers of the human tongue resolved in vivo using constrained spherical deconvolution. Journal of Magnetic Resonance Imaging 2019; 50:96–105.
47. Rehmann R, Schlaffke L, Froeling M, et al.: Muscle diffusion tensor imaging in glycogen storage disease V (McArdle disease). European Radiology 2018; 29:3224–3232.
48. Baalen S van, Froeling M, Asselman M, et al.: Mono, bi- and tri-exponential diffusion MRI modelling for renal solid masses and comparison with histopathological findings. Cancer Imaging 2018; 18:1–11.
49. Nelissen JL, Traa WA, Boer HH de, et al.: An advanced magnetic resonance imaging perspective on the etiology of deep tissue injury. Journal of Applied Physiology 2018; 124:1580–1596.
50. Oudeman J, Verhamme C, Engbersen MP, et al.: Diffusion tensor MRI of the healthy brachial plexus. PLOS ONE 2018; 13:e0196975.
51. Gurney-Champion OJ, Klaassen R, Froeling M, et al.: Comparison of six fit algorithms for the intra-voxel incoherent motion model of diffusion-weighted magnetic resonance imaging data of pancreatic cancer patients. PLOS ONE 2018; 13:e0194590.
52. Sinha S, Sinha U, Malis V, Bhargava V, Sakamoto K, Rajasekaran M: Exploration of male urethral sphincter complex using diffusion tensor imaging (DTI)‐based fiber‐tracking. Journal of Magnetic Resonance Imaging 2018; 48:1002–1011.
53. Heerden LE van, Kesteren Z van, Gurney-Champion OJ, et al.: Image Distortions on a Plastic Interstitial Computed Tomography/Magnetic Resonance Brachytherapy Applicator at 3 Tesla Magnetic Resonance Imaging and Their Dosimetric Impact. International Journal of Radiation Oncology*Biology*Physics 2017; 99:710–718.
54. Bel R van der, Gurney-Champion OJ, Froeling M, Stroes ESG, Nederveen AJ, Krediet CTP: A tri-exponential model for intravoxel incoherent motion analysis of the human kidney: In silico and during pharmacological renal perfusion modulation. European Journal of Radiology 2017; 91:168–174.
55. Schlaffke L, Rehmann R, Froeling M, et al.: Diffusion tensor imaging of the human calf: Variation of inter‐ and intramuscle‐specific diffusion parameters. Journal of Magnetic Resonance Imaging 2017; 46:1137–1148.
56. Samari B, Aumentado-Armstrong T, Strijkers G, Froeling M, Siddiqi K: Cartan frames for heart wall fiber motion. In Lecture Notes in Computer Science (including subseries Lecture Notes in Artificial Intelligence and Lecture Notes in Bioinformatics). Volume 10263 LNCS. Springer; 2017:32–41.
57. Samari B, Aumentado-Armstrong T, Strijkers G, Froeling M, Siddiqi K: Denoising moving heart wall fibers using cartan frames. In Lecture Notes in Computer Science (including subseries Lecture Notes in Artificial Intelligence and Lecture Notes in Bioinformatics). Volume 10433 LNCS. Springer; 2017:672–680.
58. Oudeman J, Mazzoli V, Marra MA, et al.: A novel diffusion‐tensor <scp>MRI</scp> approach for skeletal muscle fascicle length measurements. Physiological Reports 2016; 4:e13012.
59. Mazzoli V, Oudeman J, Nicolay K, et al.: Assessment of passive muscle elongation using Diffusion Tensor MRI: Correlation between fiber length and diffusion coefficients. NMR in Biomedicine 2016; 29:1813–1824.
60. Baalen S van, Leemans A, Dik P, Lilien MR, Haken B ten, Froeling M: Intravoxel incoherent motion modeling in the kidneys: Comparison of mono-, bi-, and triexponential fit. Journal of Magnetic Resonance Imaging 2016; 46:228–239.
61. Gurney‐Champion OJ, Bruins Slot T, Lens E, et al.: Quantitative assessment of biliary stent artifacts on MR images: Potential implications for target delineation in radiotherapy. Medical Physics 2016; 43:5603–5615.
62. Sieben JM, Otten I van, Lataster A, et al.: In Vivo Reconstruction of Lumbar Erector Spinae Architecture Using Diffusion Tensor MRI. Clinical Spine Surgery: A Spine Publication 2016; 29:E139–E145.
63. Heerden LE van, Gurney-Champion OJ, Kesteren Z van, et al.: Quantification of image distortions on the Utrecht interstitial CT/MR brachytherapy applicator at 3T MRI. Brachytherapy 2016; 15:118–126.
64. Hooijmans MT, Damon BM, Froeling M, et al.: Evaluation of skeletal muscle DTI in patients with duchenne muscular dystrophy. NMR in Biomedicine 2015; 28:1589–1597.
65. Oudeman J, Nederveen AJ, Strijkers GJ, Maas M, Luijten PR, Froeling M: Techniques and applications of skeletal muscle diffusion tensor imaging: A review. Journal of Magnetic Resonance Imaging 2015; 43:773–788.
66. Froeling M, Oudeman J, Strijkers GJ, et al.: Muscle Changes Detected with Diffusion-Tensor Imaging after Long-Distance Running. Radiology 2015; 274:548–562.
67. Froeling M, Strijkers GJ, Nederveen AJ, Chamuleau SA, Luijten PR: Diffusion Tensor MRI of the Heart – In Vivo Imaging of Myocardial Fiber Architecture. Current Cardiovascular Imaging Reports 2014; 7:1–11.
68. Froeling M, Mazzoli V, Nederveen AJ, Luijten PR, Strijkers GJ: Ex vivo cardiac DTI: On the effects of diffusion time and b-value. Journal of Cardiovascular Magnetic Resonance 2014; 16:P77.
69. Froeling M, Strijkers GJ, Nederveen AJ, Chamuleau SA, Luijten PR: Feasibility of in vivo whole heart DTI and IVIM with a 15 minute acquisition protocol. Journal of Cardiovascular Magnetic Resonance 2014; 16:O15.
70. Froeling M, Nederveen AJ, Nicolay K, Strijkers GJ: DTI of human skeletal muscle: The effects of diffusion encoding parameters, signal‐to‐noise ratio and t2 on tensor indices and fiber tracts. NMR in Biomedicine 2013; 26:1339–1352.
71. Zijta FM, Froeling M, Nederveen AJ, Stoker J: Diffusion tensor imaging and fiber tractography for the visualization of the female pelvic floor. Clinical Anatomy 2012; 26:110–114.
72. Zijta FM, Lakeman MME, Froeling M, et al.: Evaluation of the female pelvic floor in pelvic organ prolapse using 3.0-Tesla diffusion tensor imaging and fibre tractography. European Radiology 2012; 22:2806–2813.
73. Froeling M: DTI of Human Skeletal Muscle - From Simulation to Clinical Implementation. Eindhoven: Technische Universiteit Eindhoven; 2012.
74. Zijta FM, Froeling M, Paardt MP van der, et al.: Feasibility of diffusion tensor imaging (DTI) with fibre tractography of the normal female pelvic floor. European Radiology 2011; 21:1243–1249.
75. Froeling M, Oudeman J, Berg S van den, et al.: Reproducibility of diffusion tensor imaging in human forearm muscles at 3.0 T in a clinical setting. Magnetic Resonance in Medicine 2010; 64:1182–1190.
76. Dai J, Gosselink M, Shams Z, Froeling M, Raaijmakers AJE, Klomp DWJ: 31P MRSI coil combination using 23Na sensitivity information. Magnetic Resonance in Medicine.